Clinical Trial
A clinical trial is a structured research study involving human participants. Trials may test a drug, gene therapy, device, procedure, diagnostic tool, or care approach. In rare disease, trials can be especially difficult because patient populations are small, symptoms vary, natural history may be incomplete, and meaningful endpoints can be hard to measure. A trial is not the same as treatment; it is a way to gather evidence.
Why it matters
Families considering a trial need to understand the goals, risks, eligibility criteria, endpoints, follow-up, and what is known or still uncertain about the therapy being studied.
Related content
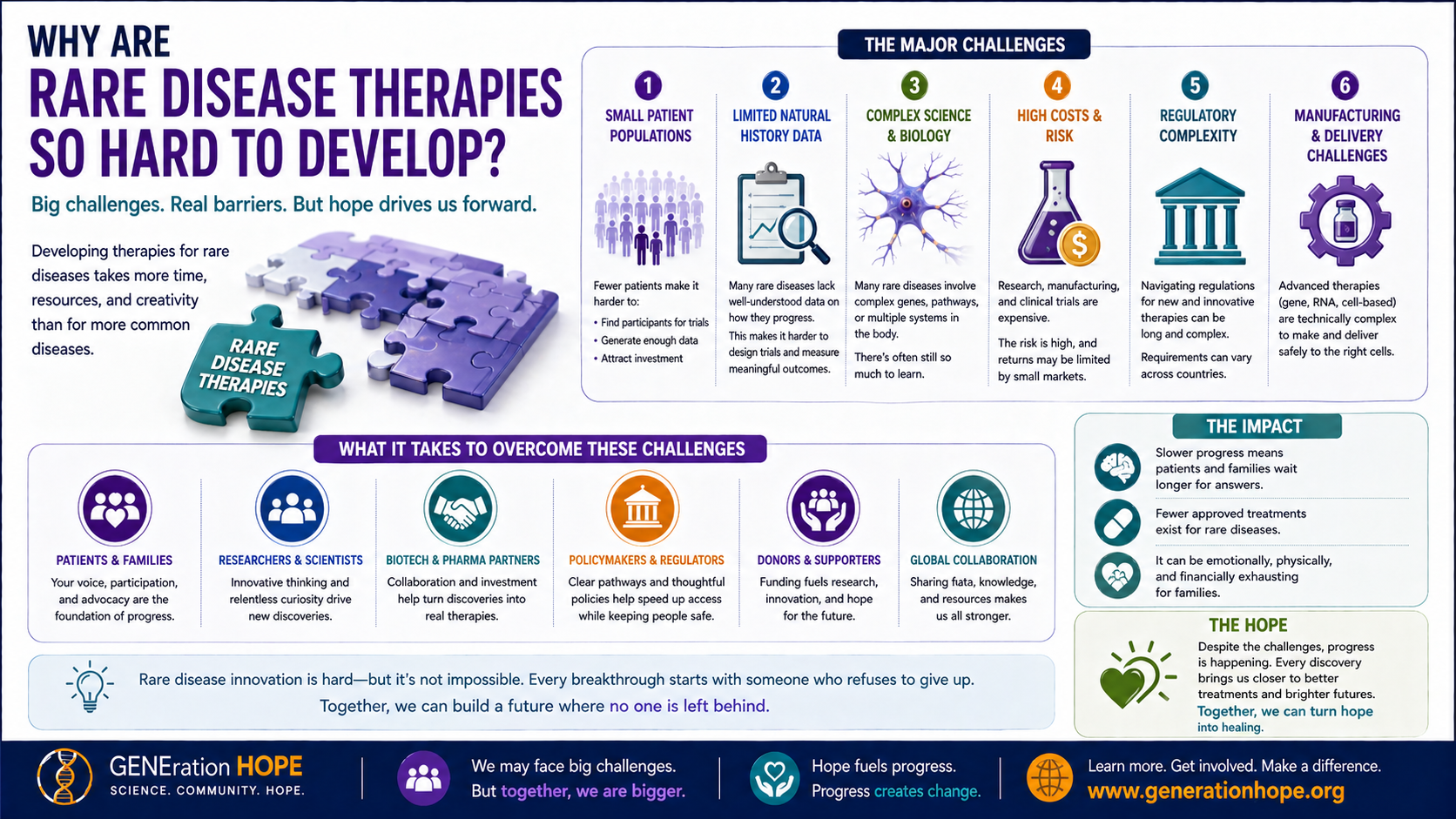
Why Are Rare Disease Therapies So Hard?
Rare disease therapies are hard because the science, trial design, manufacturing, funding, and access challenges all arrive at once.

Dr. Alex Kolevzon on Gene Therapy, SHANK3 & the Future of Treatment
Dr. Alexander Kolevzon
Welcome to Generation Hope. In this wide-ranging interview, Dr. Alex Kolevzon (Mount Sinai) sits down with Ron to discuss the science, the trials, and the human side of gene therapy for rare neurodevelopmental disorders. In this episode...
Watch InterviewHow AI Could Change Clinical Trials for Ultra-Rare Disorders
Ultra-rare trials are often small, fragile, and difficult to interpret. AI may help researchers design studies that are more realistic without lowering the bar for evidence.
Why it matters: Ultra-rare trials need rigorous design even when traditional large studies are difficult or impossible.
Source: GENEration Hope editorial analysis
Read