Phase I/II
Phase I/II is a trial label often used when a study combines goals from early phases of clinical research. Phase I usually focuses on safety, tolerability, dose, and how the therapy behaves in the body. Phase II begins to look more directly at whether the therapy has biological activity or possible benefit. In rare disease, phases are sometimes blended because patient populations are small and every participant's data matters.
Why it matters
Families should know that a Phase I/II trial may offer hope, but its main purpose is still to gather evidence. Early signals are important, but they are not the same as proof that a therapy works.
Related content
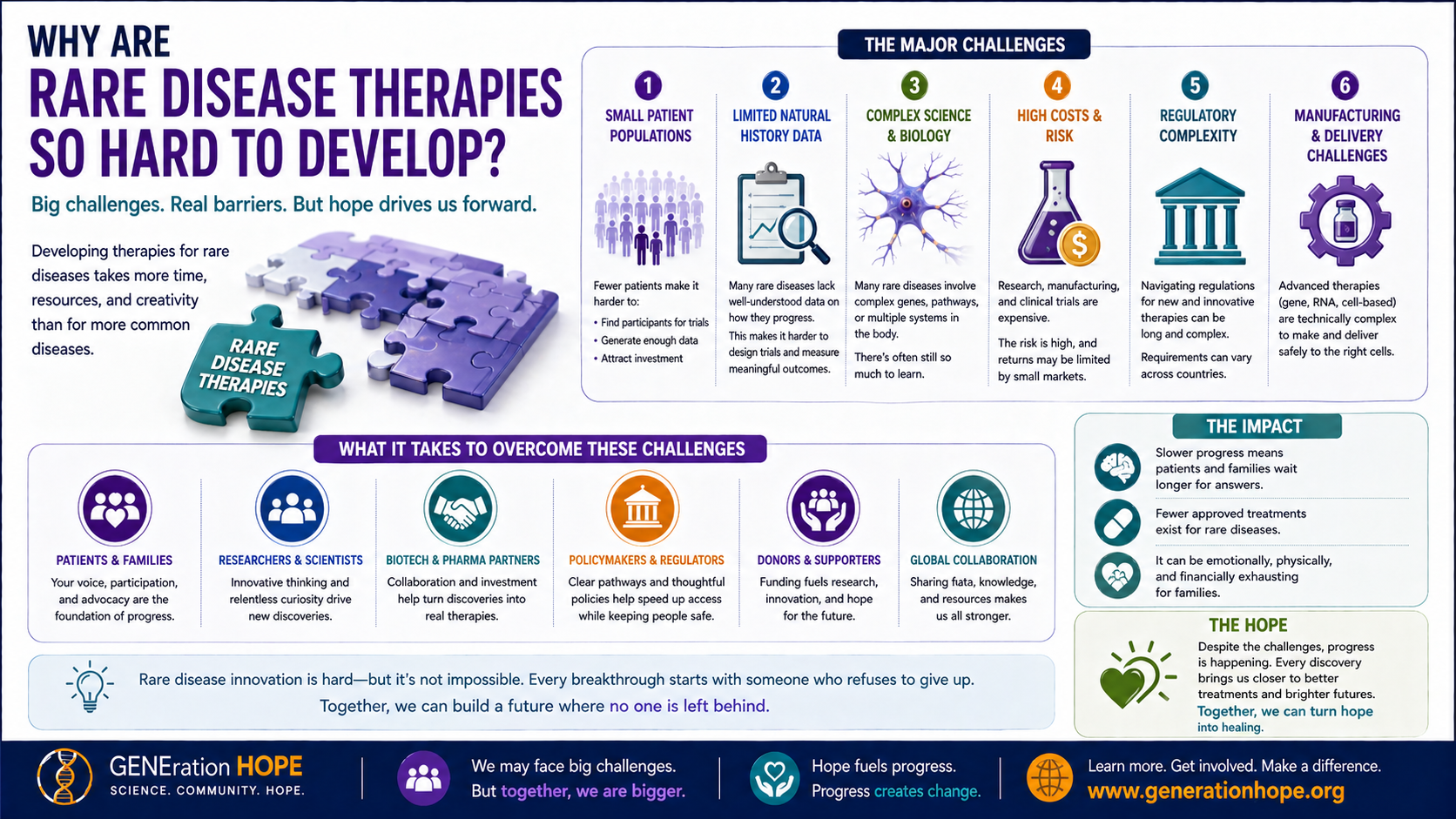
Why Are Rare Disease Therapies So Hard?
Rare disease therapies are hard because the science, trial design, manufacturing, funding, and access challenges all arrive at once.

Dr. Alex Kolevzon on Gene Therapy, SHANK3 & the Future of Treatment
Dr. Alexander Kolevzon
Welcome to Generation Hope. In this wide-ranging interview, Dr. Alex Kolevzon (Mount Sinai) sits down with Ron to discuss the science, the trials, and the human side of gene therapy for rare neurodevelopmental disorders. In this episode...
Watch InterviewHow AI Could Change Clinical Trials for Ultra-Rare Disorders
Ultra-rare trials are often small, fragile, and difficult to interpret. AI may help researchers design studies that are more realistic without lowering the bar for evidence.
Why it matters: Ultra-rare trials need rigorous design even when traditional large studies are difficult or impossible.
Source: GENEration Hope editorial analysis
Read