Inside the Fight to Cure Rett Syndrome – Monica Coenraads Interview
Monica Coenraads
Founder and CEO, Rett Syndrome Research Trust
Monica Coenraads, founder and CEO of the Rett Syndrome Research Trust (RSRT), has led the charge to cure Rett Syndrome—raising over $123 million and driving multiple gene therapy programs now in clinical trials. In this in-depth conversation, Monica shares her personal journey from her daughter Chelsea’s diagnosis to building one of the world’s most impactful rare disease research organizations. She explains the evolution of Rett research—from the discovery of MECP2 to today’s cutting-edge genetic medicines including gene therapy, base and prime editing, and RNA trans-splicing. We discuss: • The founding of RSRT and early breakthroughs • The reversibility discovery that changed everything • The Taysha and Neurogene gene therapy trials • The future of Rett research (Roadmap to Cures 2.0) • How parents can catalyze scientific progress • The power of early diagnosis and newborn screening Stay tuned after the interview for updates on the latest Rett, STXBP1, and Phelan-McDermid Syndrome gene therapy trials. 👉 Subscribe for more interviews with scientists, researchers, and advocates advancing gene therapy for rare genetic disorders. 🌐 Visit GENErationHope.co for global updates and resources. 🕒 Chapters / Timestamps 00:00 – Introduction 00:28 – About Monica Coenraads & RSRT 01:53 – Chelsea’s Diagnosis and Early Signs 05:04 – Discovering MECP2 and Rett Syndrome’s Cause 07:22 – Founding the Rett Syndrome Research Foundation 08:29 – The Breakthrough Reversibility Experiment 11:10 – Turning to Gene Therapy (2008–2010) 13:08 – Parental Perspective: Asking Naive but Important Questions 15:23 – What is Rett Syndrome? Symptoms and Progression 18:13 – Prevalence and Underdiagnosis: Newborn Screening Data 22:07 – Diagnostic Delays and Barriers for Families 25:58 – Founding RSRT: A Focus on Genetic Medicines 27:24 – Major Milestones in 25 Years of Rett Research 30:31 – Trials and Setbacks: Novartis, Avexis, and Lessons Learned 32:20 – Advice for Newly Diagnosed Families 36:22 – How RSRT Raised $100 Million Through Families 38:05 – Why Funding Still Matters Most Today 39:13 – RSRT’s Team and Family-Driven Urgency 44:07 – The Importance of Early Diagnosis 47:05 – Debating Newborn Screening Policy 48:41 – The Current Gene Therapy Trials (Taysha & Neurogene) 53:29 – Roadmap to Cures 2.0: What’s Next for Rett Research 57:09 – The Future: Base Editing, Prime Editing, and RNA Approaches 1:00:27 – The Challenges of Delivery (AAV9 and Beyond) 1:02:05 – Ranking the Modalities & FDA Collaboration 1:07:32 – Collaboration Across Rare Disease Communities 1:08:47 – Final Thoughts & Message to Families 1:09:30 – Closing Monologue: Updates on Rett & STXBP1 Trials
Key topics
- Rett research
- MECP2
- gene therapy trials
- foundation strategy
Chapters
- 1. – Introduction 00:28 – About Monica Coenraads & RSRT 01:53 – Chelsea’s Diagnosis and Early Signs 05:04 – Discovering MECP2 and Rett Syndrome’s Cause 07:22 – Founding the Rett Syndrome Research Foundation 08:29 – The Breakthrough Reversibility Experiment 11:10 – Turning to Gene Therapy (2008–2010) 13:08 – Parental Perspective: Asking Naive but Important Questions 15:23 – What is Rett Syndrome? Symptoms and Progression 18:13 – Prevalence and Underdiagnosis: Newborn Screening Data 22:07 – Diagnostic Delays and Barriers for Families 25:58 – Founding RSRT: A Focus on Genetic Medicines 27:24 – Major Milestones in 25 Years of Rett Research 30:31 – Trials and Setbacks: Novartis, Avexis, and Lessons Learned 32:20 – Advice for Newly Diagnosed Families 36:22 – How RSRT Raised $100 Million Through Families 38:05 – Why Funding Still Matters Most Today 39:13 – RSRT’s Team and Family-Driven Urgency 44:07 – The Importance of Early Diagnosis 47:05 – Debating Newborn Screening Policy 48:41 – The Current Gene Therapy Trials (Taysha & Neurogene) 53:29 – Roadmap to Cures 2.0: What’s Next for Rett Research 57:09 – The Future: Base Editing, Prime Editing, and RNA Approaches 1:00:27 – The Challenges of Delivery (AAV9 and Beyond) 1:02:05 – Ranking the Modalities & FDA Collaboration 1:07:32 – Collaboration Across Rare Disease Communities 1:08:47 – Final Thoughts & Message to Families 1:09:30 – Closing Monologue: Updates on Rett & STXBP1 Trials
Transcript
Transcript coming soon. This page is already connected to the published YouTube interview, and the edited transcript can be added here for search and accessibility.
Related explainers and articles
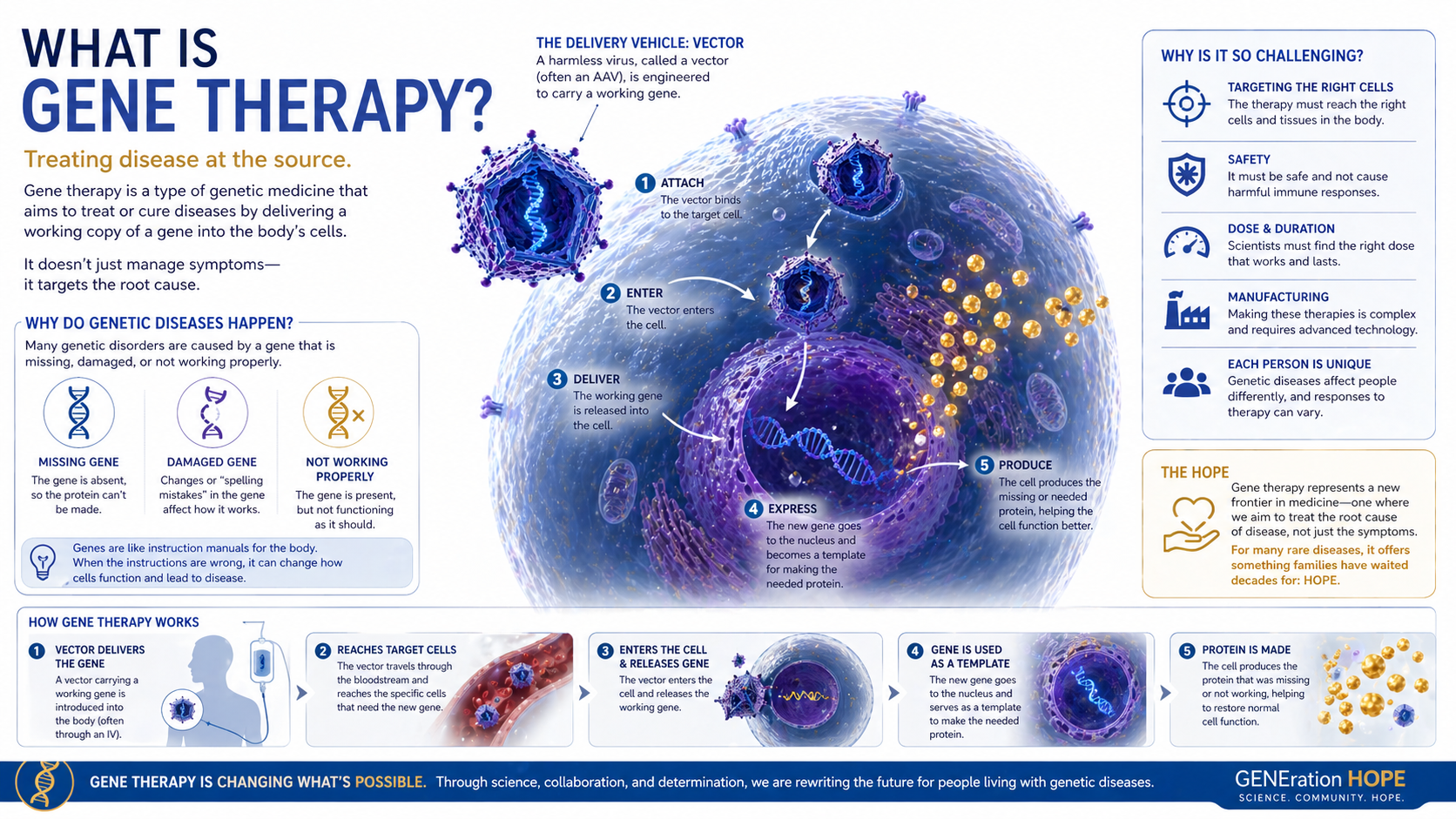
What Is Gene Therapy?
Gene therapy is a treatment approach that tries to address disease at the level of the gene, often by adding, replacing, silencing, or editing genetic instructions.
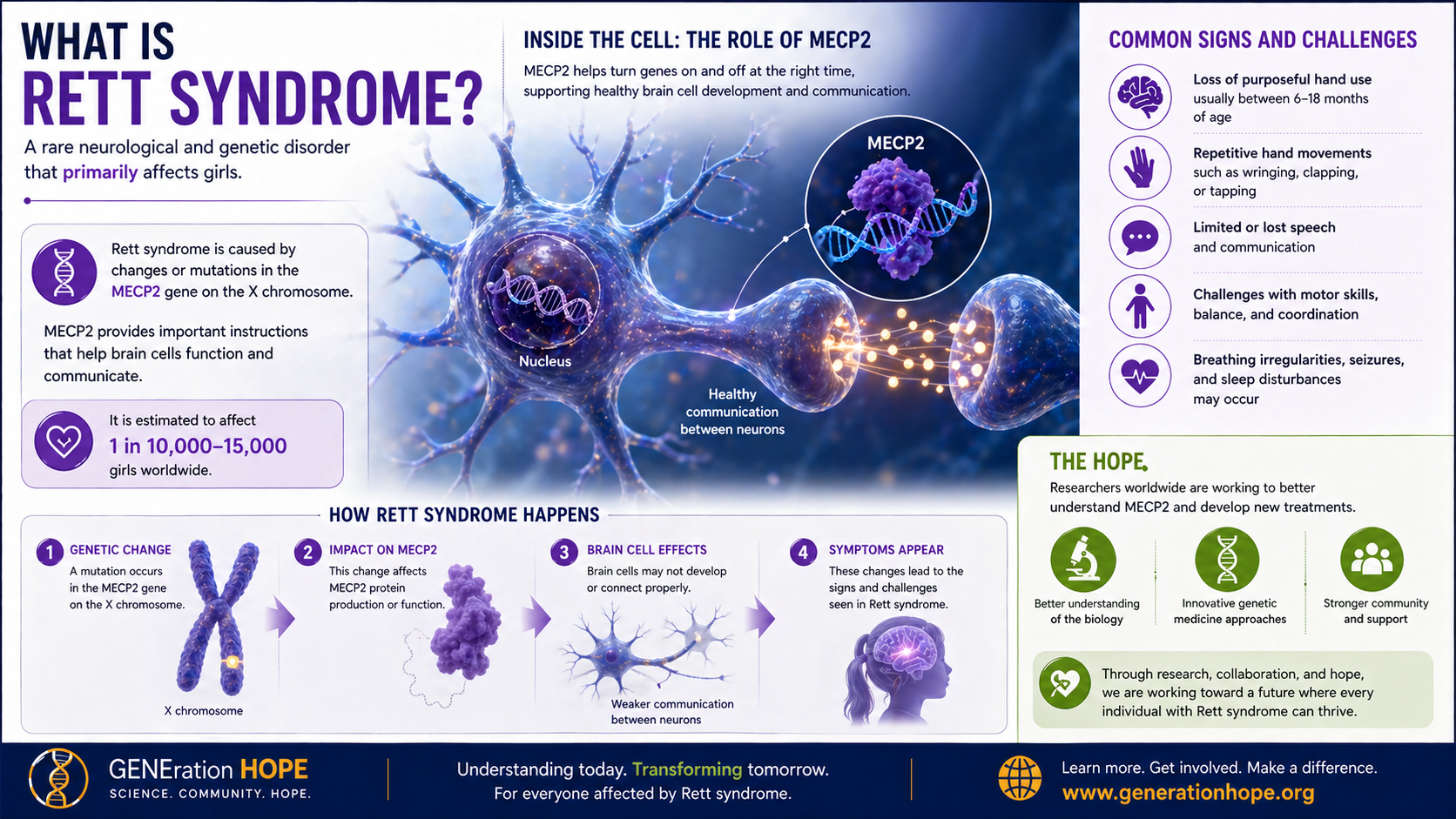
What Is Rett Syndrome?
Rett syndrome is a rare neurodevelopmental disorder, most often linked to MECP2, that usually appears after early development seems relatively typical.
Related interviews

Dr. Guoping Feng (MIT) on SHANK3 Gene Therapy, Brain Disorders, and What’s Coming Next
Dr. Guoping Feng
Welcome — I’m Ron Kleiman, and this is GENEration Hope. In this episode, I’m joined by Dr. Guoping Feng, Professor of Neuroscience at MIT, affiliated with the McGovern Institute, the Yang Tan Collective, and the Broad Institute. 
Watch Interview
How a Parent-Led Movement Grows: Sue Lomas (Phelan-McDermid Syndrome Foundation)
Sue Lomas
Sue Lomas helped build the Phelan-McDermid Syndrome community in the earliest days—when families were scattered, information was scarce, and finding “your people” took persistence. In this conversation, Sue and I talk about how rare-dise...
Watch Interview